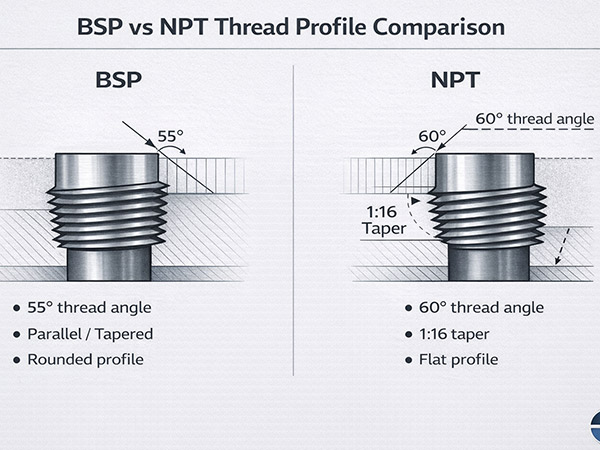
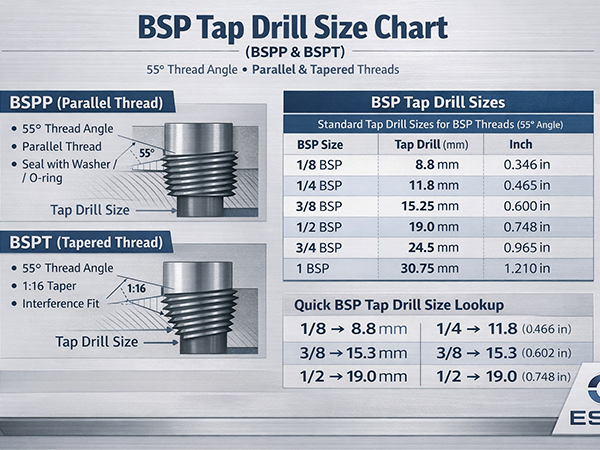
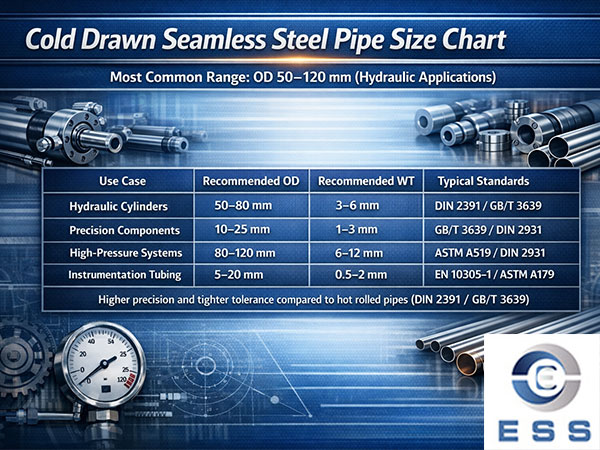
The main reason for rusting of mild steel
pipes is due to the oxidation reaction between their surface and oxygen and
moisture in the air. The carbon
steel pipe generally contains iron elements, which are easily oxidized to
form iron oxide (Fe2O3) and iron hydroxide (Fe(OH)2), which causes red rust on
the surface of mild steel pipes.
What causes mild steel pipe to rust
1. Oxygen and moisture in the air
After the surface of the mild steel pipe
comes into contact with oxygen and moisture in the air, an oxidation reaction
occurs, resulting in rust. Like common seamless
steel pipe, in underground pipe corridors with poor ventilation and high
humidity, oxidation and rust are very likely to occur due to long-term contact
with humid air.
2. Environmental factors
If there are certain acidic and alkaline
substances in the environment, it may also cause rusting of mild steel pipes.
In addition, if exposed to a humid environment for a long time, it is also easy
to cause rusting of mild steel pipes. For example, in the production area of chemical enterprises, some carbon steel pipes that transport acidic
or alkaline media are affected by the volatilization or leakage of chemicals,
which accelerates the rusting process of the pipes.
3. Improper surface treatment
Improper surface treatment of mild steel
pipes, such as unclean cleaning, grease pollution, surface indentation, etc.,
are easy to cause rusting of ms pipes. In some factories, after welding with pipe flange, the
welding residues are not cleaned up, which becomes the starting point of rust.
Why does mild steel rust easily
The main component of rust on mild steel is
iron oxide. In humid environments, the iron on the surface of mild steel reacts
with oxygen and water in the air to form hydrated iron(III) oxides, which is
what we commonly call "rust."
The main chemical component of rust is iron
oxide (Fe2O3). It is reddish-brown, porous, and easily peels off. Compared to
the original steel material, its strength and toughness are significantly
reduced.
Mild steel has a low carbon content, only
0.05%-0.25%, making it more susceptible to corrosion from moisture, fog, acidic
and alkaline environments compared to other steels, thus leading to rusting.
Simultaneously, the iron in mild steel also reacts with oxides in the air to
form red rust, further exacerbating its susceptibility to rust.
The rust formation process of mild steel
The rusting process of mild steel is a
typical electrochemical corrosion process.
In a humid environment, a thin film of
water forms on the surface of steel. Dissolved oxygen in the water reacts with
the iron in a redox reaction. Iron, acting as the anode, is oxidized to ferric
ions (Fe²⁺), while oxygen
is reduced to hydroxide ions (OH⁻) at the cathode. The two combine to form ferrous hydroxide (Fe(OH)₂).
Subsequently, ferrous hydroxide is further
oxidized to ferric hydroxide (Fe(OH)₃), and finally dehydrates to form iron oxide (Fe₂O₃), i.e., rust.
Differences in rusting between mild steel
and high-carbon steel
1. Differences in Chemical Composition
The biggest difference between mild steel
and high-carbon steel lies in their carbon content. Mild steel has a carbon
content below 0.05%, while high-carbon steel has a carbon content above 0.6%.
Besides this, there are other differences in the chemical composition of the
two types of steel.
2. Rusting Mechanism
Due to its lower carbon content, mild steel
readily reacts with oxygen when exposed to air, moisture, and other organic
matter. When the surface of mild steel is moist, the iron on the surface is
oxidized and converted into iron oxides, i.e., rust.
High-carbon steel has a higher carbon
content, resulting in a higher chromium content. Chromium is a natural oxide
that forms a dense chromium oxide layer on the steel surface, effectively
preventing rust.
3. Rust Prevention Methods
For mild steel, rust can be prevented or
treated by applying rust inhibitors such as paint, coatings, or iron oxide to
the steel surface; increasing the steel's rust resistance level, such as by
zinc plating after surface treatment or adding other corrosion-resistant elements
like copper, nickel, or molybdenum.
For high-carbon steel, a simpler method is
sufficient: keep the surface clean and dry. If slight rust appears, gently wipe
it with sandpaper or a steel brush.
How to avoid rusting of mild steel pipes
1. Control environmental conditions
Try to store mild steel pipes in a dry,
ventilated, and weathering-free environment to avoid the invasion of mild steel
pipes by moisture, acidic and alkaline substances.
2. Surface treatment
For the treatment of the surface of mild
steel pipes, it is necessary to ensure that the surface is clean, remove all
impurities such as grease, steel slag, etc., and take anti-corrosion measures,
such as galvanizing and chrome plating.
3. Packaging and storage
During production, transportation and
storage, attention should be paid to the packaging of mild steel pipes to
prevent mild steel pipes from being affected by the external environment. At
the same time, when storing, it should be stored in a dry, ventilated and
temperature-appropriate place to avoid the mild steel pipe from being attacked
by a humid environment.
Conclusion
The above is a detailed introduction to the
causes of rusting of mild steel pipes and preventive measures. In the process
of using mild steel pipes, it is necessary to pay attention to the surface
treatment of mild steel pipes, control the storage environment, and avoid the
mild steel pipes from being attacked by moisture, acidic and alkaline
substances. As long as it is stored and used in the correct way, the problem of
rusting of mild steel pipes can be effectively avoided.
Read more: Difference between mild steel pipe and carbon steel pipe or How long can mild steel pipes last?













 Eastern Steel Manufacturing Co.,Ltd not only improve product production and sales services, but also provide additional value-added services. As long as you need, we can complete your specific needs together.
Eastern Steel Manufacturing Co.,Ltd not only improve product production and sales services, but also provide additional value-added services. As long as you need, we can complete your specific needs together.