1. What Is the Purpose of Pickling Seamless Steel Pipes?
Pickling and passivation of seamless steel pipes is a chemical reaction process.
First, the oxide scale and rust on the steel surface are removed through a pickling solution, and the passivation process is completed at the same time.
It can effectively prevent the oxidation of seamless steel pipes and achieve the purpose of anti-corrosion.
Before pickling and passivating seamless steel pipes, oil, degreasing residues, surface drawing compounds, and other impurities must be removed.
Before cold drawing, surface treatment such as pickling and lubrication is essential. Learn more about cold drawn seamless steel pipe.
After pickling and passivation, the surface of the seamless steel pipe turns into a uniform silvery white color, which greatly improves the corrosion resistance of stainless steel.

2. Why Pickling Is Necessary for Seamless Steel Pipes
During the process of prefabrication, welding, testing, and heat treatment, iron oxide, welding slag, grease, and other dirt will accumulate on the surface of the pipe (including carbon steel pipe and stainless steel pipe), causing the corrosion resistance of the pipe to change.
After the pipe is cleaned with pickling liquid, the dirt on the surface can be removed. Through passivation treatment, a protective film against oxidation can be formed on the surface of the steel pipe, improving corrosion resistance and ensuring normal production processes.
Improper pickling may lead to surface defects during processing. See common issues in seamless pipes.
If oxide scale is not removed before cold processing:
The hardness of the oxide scale is higher than the base metal
It will be pressed into the pipe surface during deformation
Friction increases and tool wear intensifies
Surface quality decreases
As a result, the cold working process cannot proceed effectively.
Therefore, removing oxide scale on the inner and outer surfaces of seamless steel pipes is a critical step in production.

3. Key Functions of Pickling
3.1 Removal of Oxide Scale and Rust
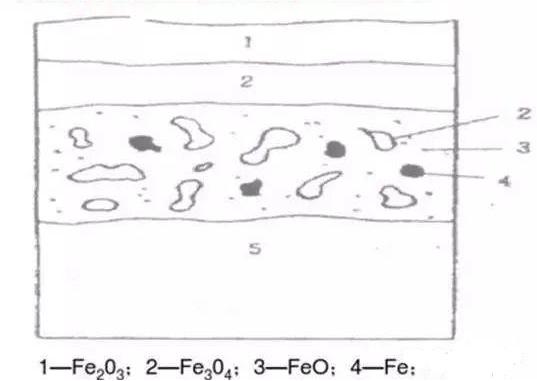
Pickling uses acid solutions to chemically remove iron oxide scale (Fe₂O₃, Fe₃O₄, FeO) from the metal surface.
3.2 Improvement of Corrosion Resistance
After pickling, passivation forms a thin, dense protective film on the surface, preventing the metal from contacting corrosive media.
3.3 Surface Cleaning and Quality Enhancement
Pickling removes welding slag, oil, grease, and other contaminants, resulting in a clean and uniform surface.
3.4 Preparation for Further Processing
Pickling is an essential step before:
Cold drawing
Phosphating
Coating or painting
Lubrication treatment
It ensures better adhesion, reduced friction, and improved processing performance.
4. Pickling Principle of Seamless Steel Pipes
4.1 Definition of Pickling
Acids use chemical methods to remove iron oxide scale under a certain concentration, temperature, and reaction rate. This process is called pickling.
4.2 Principle of Pickling Reaction
Pickling is essentially a chemical process used to remove iron oxide scale from the metal surface, so it is also called chemical pickling.
The iron oxide scale formed on the surface of steel pipes mainly includes Fe₂O₃, Fe₃O₄, and FeO. These oxides are insoluble in water but can react with acid solutions. When immersed in acid or when acid is applied to the surface, a series of chemical reactions occur:
(1) Dissolution of iron oxide scale
Iron oxide reacts with acid and is dissolved.
(2) Reaction between metal and acid (hydrogen evolution)
Metal iron reacts with acid to generate hydrogen gas, which produces a mechanical peeling effect on the oxide scale.
(3) Reduction reaction
The generated atomic hydrogen reduces iron oxide into ferrous oxide, which is easier to react with acid and be removed.
4.3 Structural Characteristics of Oxide Scale
The iron oxide scale on ordinary carbon steel seamless pipes is mainly composed of three layers:
Outer layer: Fe₂O₃ (highest oxygen content)
Middle layer: Fe₃O₄
Inner layer: FeO (closest to the metal substrate)
Under temperatures above 575°C, the oxide scale structure typically shows a three-layer distribution.
The inner layer is the thickest (about 80%–85%), while the outer layer is the thinnest.
Because the oxide scale is loose, porous, and often cracked, acid solution can penetrate through these defects and react not only with the oxide layer but also with the base metal.
4.4 Pickling Classification (Condensed)
According to different conditions, pickling can be classified as:
(1) By acid type:
sulfuric acid pickling, hydrochloric acid pickling, nitric acid pickling, hydrofluoric acid pickling
(2) By material:
carbon steel uses sulfuric or hydrochloric acid,
stainless steel usually uses nitric acid + hydrofluoric acid mixed solution
(3) By process/equipment:
tank pickling, semi-continuous pickling, continuous pickling
5. Passivation of Seamless Steel Pipes
5.1 Passivation Principle
The passivation mechanism is generally explained by thin film theory.
When metal reacts with oxidizing substances, a very thin but dense film forms on the surface.
This film is not simply a deposit — it is tightly bonded to the metal and exists as a stable oxide layer. Once formed, it effectively separates the base metal from air and moisture.
In practical terms, the metal surface enters a “passive state”, meaning the corrosion reaction is greatly slowed down rather than completely stopped.
5.2 Characteristics of the Passivation Film
In actual production, the passivation film on seamless steel pipes shows several typical features.
It is dense and relatively stable, with good coverage on the metal surface.
At the same time, it can isolate air and moisture, which is the key reason for improved corrosion resistance.
Another important characteristic is its self-repairing ability.
If the film is slightly damaged, it can reform in air under suitable conditions, which is something traditional anti-rust oil cannot achieve.
5.3 Practical Advantages of Passivation
Compared with conventional protection methods (such as oil coating), passivation is more suitable for industrial processing:
It does not increase the thickness of the workpiece and does not affect dimensional accuracy
The surface appearance remains basically unchanged
The solution can be reused in many cases, so the cost is relatively controllable
The resulting protection is more stable, especially for long-term storage or transportation
5.4 Why Pickling Is Always Followed by Passivation
In actual production, pickling and passivation are rarely separated.
Pickling removes oxide scale, welding residues, and surface contamination, exposing fresh metal.
However, this freshly exposed surface is also more reactive and prone to re-oxidation.
This is where passivation becomes necessary.
By forming a protective film immediately after pickling, the surface condition is stabilized, and the risk of secondary oxidation is reduced.
For this reason, the two processes are usually treated as a combined step in seamless steel pipe manufacturing.
6. Phosphating and Saponification of Seamless Steel Pipes
6.1 Phosphating Process
Phosphating is a typical chemical surface treatment used after pickling.
The steel pipe is immersed in a phosphating solution, where a chemical reaction takes place and a phosphate conversion film forms on the surface.
This film is crystalline in structure and insoluble in water.
Unlike passivation, it is not just for corrosion resistance — it also serves as a functional layer for subsequent processing.
6.2 What Phosphating Is Actually Used For
In real applications, phosphating is not used for a single purpose.
Its role depends on the downstream process:
As a protective layer, it helps isolate the metal from the environment
As a pre-coating treatment, it significantly improves paint adhesion
In cold working, it acts as a carrier layer for lubrication
It can also improve the uniformity of the surface appearance
Because of this versatility, phosphating is widely used in industries such as automotive, machinery, and pipeline manufacturing.
6.3 Saponification (Lubrication Step After Phosphating)
After phosphating, a saponification treatment is often applied, especially for pipes that will undergo drawing or forming.
At this stage, the phosphated pipe is immersed in a saponification solution.
The solution reacts with the phosphating film and forms a lubricating layer on top of it.
In practice, this step is critical for cold working:
It reduces friction during drawing or stretching
Helps protect the pipe surface from damage
Improves tool life and process stability
Typically, the phosphating film thickness is around 8–12 μm before entering this stage.
6.4 How These Processes Work Together
In production lines, these treatments are not isolated steps but part of a continuous sequence.
A typical process looks like this:
Pickling → Washing → Phosphating → Washing → Saponification → Drying
For cold drawing applications, the sequence is more focused:
Pickling → Phosphating → Saponification → Cold drawing
Each step prepares the surface for the next one, which is why skipping or simplifying any stage can directly affect the final product quality.
Still have questions about seamless steel pipe surface treatment? Here are some common questions from engineers and buyers.
7. FAQs
Q1. Why is pickling required before cold drawing?
Pickling removes hard oxide scale from the pipe surface.
Without it, friction increases, surface defects occur, and tool wear becomes severe during cold drawing.
Q2. What acids are used for pickling steel pipes?
Carbon steel: hydrochloric acid or sulfuric acid
Stainless steel: nitric acid + hydrofluoric acid
The selection depends on the material and process requirements.
Q3. What is the difference between pickling and passivation?
Pickling removes oxide scale and contaminants.
Passivation forms a protective film to prevent corrosion.
In practice, pickling cleans the surface, and passivation protects it.
Q4. Is pickling always followed by passivation or phosphating?
Not always. Passivation is used for corrosion resistance, while phosphating is used for coating or cold forming processes.
Q5. Does pickling affect pipe dimensions?
Normally, no.
Pickling only removes a very thin surface layer, but over-pickling can cause slight material loss if not controlled.
Read more: Seamless Steel Pipe Sizes and Stainless steel seamless pipe vs Seamless carbon steel pipe













 Eastern Steel Manufacturing Co.,Ltd not only improve product production and sales services, but also provide additional value-added services. As long as you need, we can complete your specific needs together.
Eastern Steel Manufacturing Co.,Ltd not only improve product production and sales services, but also provide additional value-added services. As long as you need, we can complete your specific needs together.