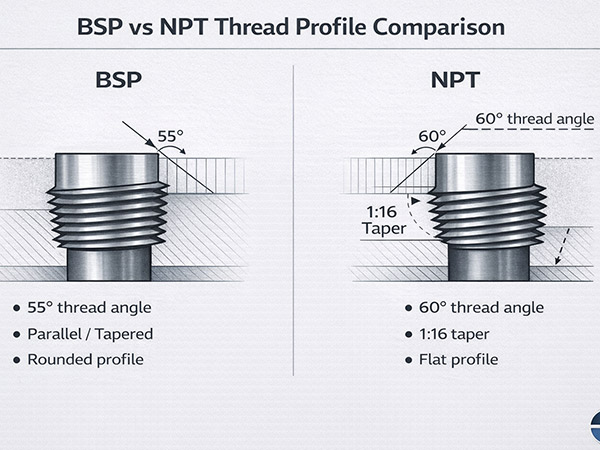
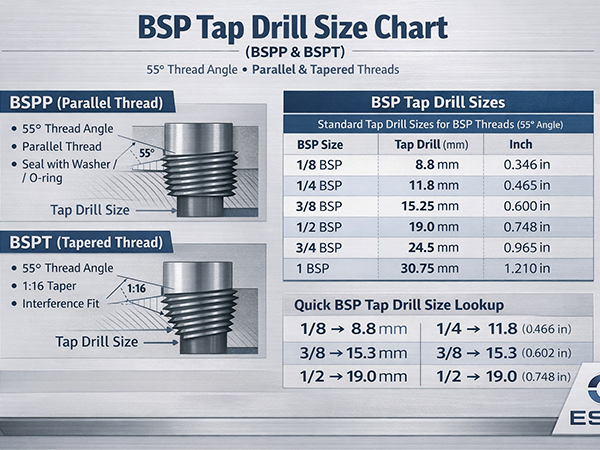
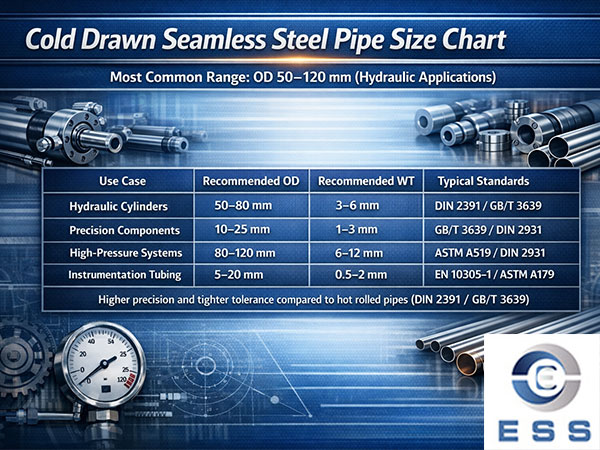
How to prevent surface decarburization of seamless steel pipes?
The carbon element in the seamless pipe has a great influence on its performance, so when the seamless pipe is decarbonized, its service life will be greatly reduced. Moreover, if the decarburized layer on the surface of the seamless pipe is not removed, the hardness and wear resistance of the surface of the seamless pipe will be reduced, and cracks will appear on the surface of the workpiece due to the volume change of the inner and outer layers during quenching. Therefore, how to prevent the surface decarburization of seamless pipes is a problem that should be paid attention to in the production process.

The heat treatment process is the key to solving the problem of surface decarburization. Because the decarburization and the oxidation of the steel wire are carried out at the same time, as long as the seamless pipe is in contact with the air as little as possible during the heat treatment process, the purpose of improving the decarburization can be achieved. The production process is as follows: the first molten lead tank plays the role of austenitizing the steel wire. The lead liquid is heated to 871°C in advance, the steel wire is austenitized through the lead liquid, and then enters the oil tank for quenching, and then enters the second Molten lead bath tempered (482°C). Because the steel wire is not in contact with air in molten lead, decarburization is effectively avoided. How to prevent lead dust pollution The main problem solved is how to prevent lead dust pollution.
The atmosphere for decarburization on the surface of the
ms seamless tube is mainly oxidizing gases such as oxygen, water vapor and carbon dioxide. When these oxidizing gases contact the surface of the heated steel wire, oxidation and decarburization occur simultaneously; the solid solution carbon in iron has a greater affinity with these gases, and the surface carbon is removed first. The above gases are generally brought in from outside the furnace; the iron oxide scale, rust on the surface of the seamless pipe and the residue on the surface of the steel wire after cold drawing will decompose after heating in the furnace, and some oxidizing gases will be generated by the reaction.
The purposeful control of the furnace atmosphere to make it in a reduced state can effectively avoid surface decarburization. For example, by controlling the ratio of carbon dioxide and carbon monoxide in the furnace, neither oxidation nor decarbonization occurs at the equilibrium point; when the proportion of carbon dioxide exceeds the equilibrium point, oxidative decarbonization occurs; when it is lower than the equilibrium point, oxidative decarbonization does not occur. The specific value of the equilibrium point should be calculated based on the carbon content of the iron and the temperature. In addition, spraying neutral protective nitrogen into the furnace is also an effective measure. At the same time, the positive pressure in the furnace can be maintained, air infiltration can be prevented, and decarburization of steel parts can be reduced or avoided.













 Eastern Steel Manufacturing Co.,Ltd not only improve product production and sales services, but also provide additional value-added services. As long as you need, we can complete your specific needs together.
Eastern Steel Manufacturing Co.,Ltd not only improve product production and sales services, but also provide additional value-added services. As long as you need, we can complete your specific needs together.